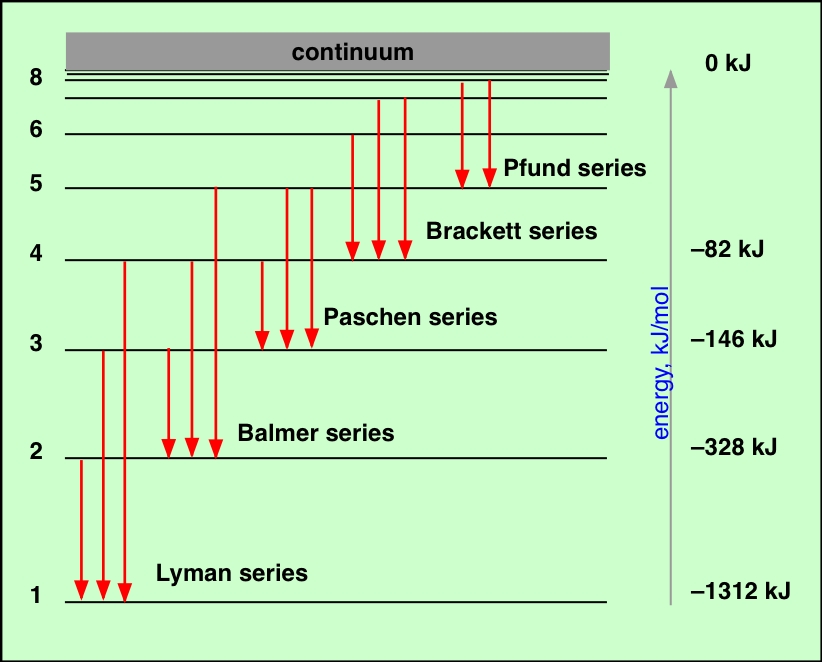 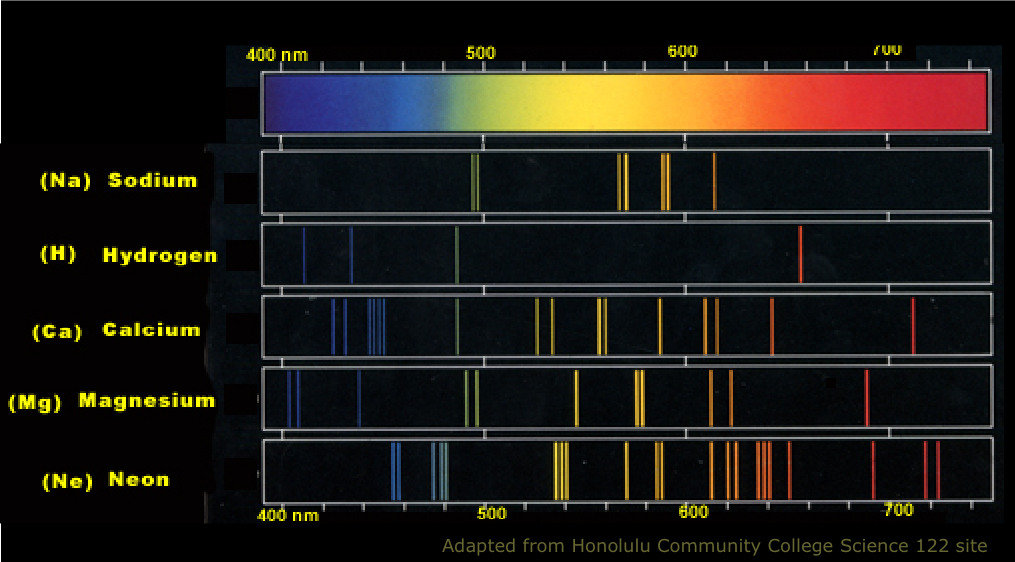
This energy is equal the difference between the. Each of these energy packets is known as a quantum (plural: quanta). In 1913, Danish physicist Niels Bohr applied Max Planck’s quantum theory to the nuclear atom of Ernest Rutherford, thus formulating the well-known planetary model of the atom, wherein electrons orbit a central nucleus in well-defined levels of energy ( Figure 1 ). To the electron fall back, it emits an amount of energy in form of light (photon) with a specific wavelength. The reality, which Planck discovered, is that energy is not continuous but quantized-meaning that it can only be transferred in individual “packets” (or particles) of the size h ν h\nu h ν h, \nu. If the kinetic energy of each electron in the beam is above a few electronvolts, collisions with atoms in the gas result in the transfer of energy to the atoms. This was a shocking discovery, because it challenged the idea that energy was continuous, and could be transferred in any amount. Emission spectra For this relationship: As the energy levels have different values, each of the possible electron transitions within an atom will produce a. 6 2 6 × 1 0 − 3 4 J ⋅ s 6, point, 626, times, 10, start superscript, minus, 34, end superscript, start text, space, J, end text, dot, start text, s, end text, and ν \nu ν \nu is the frequency of the light absorbed or emitted. Atoms and Light Exploring Atomic and Electronic Structure Unit Overview This unit covers the progression of the model of the atom from the discovery of the first sub- atomic particle to the development of quantum mechanics. There was evidence for internal structure in atoms, in the specific, unchanging frequencies of emitted and absorbed light for different atoms. Planck observed that matter actually absorbed or emitted energy only in whole-number multiples of the value h ν h\nu h ν h, \nu, where h h h h is Planck’s constant, 6.626 × 1 0 − 34 J ⋅ s 6.626\times10^ 6. Atoms and the electromagnetic spectrum were linked: atoms emitted and absorbed light, and could be distinguished by their spectra (the pattern of emitted and absorbed light of different frequencies). Planck found that the electromagnetic radiation emitted by blackbodies could not be explained by classical physics, which postulated that matter could absorb or emit any quantity of electromagnetic radiation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
